Shares of Epizyme, Inc. EPZM have plunged 58.3% in the past three months compared with the industry’s decrease of 22.4%.
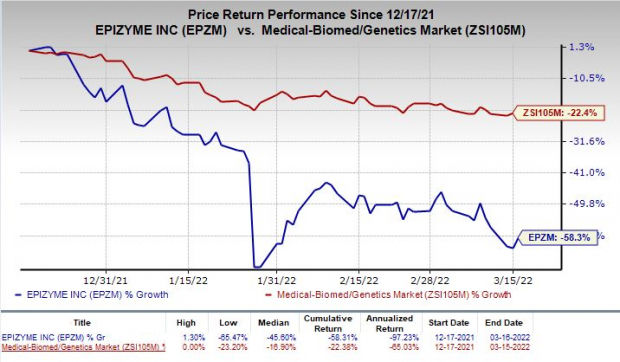
Image Source: Zacks Investment Research
In January 2022, the company floated a secondary issue of 56,666,667 shares of its common stock to the public at an issue price of $1.50 per share (excluding underwriting discounts), approximately amounting to $85 million. Epizyme also granted an option to underwriters of the issuance to purchase an additional 8.5 million shares at the same price.
Epizyme’s stock took a sharp fall as a result of this news, which can also cause the stock price to remain weak in this time frame. The fall in share price was likely due to the issuance of a large number of shares, which dilutes the company’s current shareholder base.
Epizyme plans to use the net proceeds from this new issue combined with its existing cash balance to fund the clinical studies (both ongoing and planned) of its pipeline candidates and accelerate the commercial adoption of its only marketed drug, Tazverik, which is approved for the treatment of certain patients with epithelioid sarcoma (“ES”) and follicular lymphoma (“FL”).
In 2020, tazemetostat was granted approval by the FDA under an accelerated pathway to treat ES and FL indications. Tazemetostat is marketed by Epizyme under the trade name Tazverik, which is currently the only FDA-approved drug in the company’s portfolio of marketed drugs.
Since the FDA approval for Tazverik to treat both indications is under accelerated pathway, the company is also conducting confirmatory studies for ES and FL with Tazverik. A phase Ib/III confirmatory study (EZH-301) is evaluating Tazverik in combination with doxorubicin compared with doxorubicin plus placebo as a front-line treatment for ES.
Another phase Ib/III confirmatory study (EZH-302, SYMPHONY-1) is evaluating Tazverik in combination with R2 (the combination of Revlimid plus Rituxan) compared with R2 plus placebo for treating patients with relapsed or refractory follicular lymphoma (R/R FL) previously treated with at least one systemic therapy, including those who are rituximab-refractory and/or have experienced progression of disease within two years.
In March 2022, Epizyme dosed the first patient in the phase III portion of the SYMPHONY-1 (EZH-302) study.
Also, during the fourth quarter of 2021, the company initiated a phase Ib/II basket study, EZH-1501, to evaluate the safety and efficacy of tazemetostat across multiple new types of hematological malignancies. The company has already initiated a bispecific cohort of the study evaluating a combination of tazemetostat with Roche’s RHHBY mosunetuzumab in patients with R/R FL who have received two or more prior lines of therapy. Preliminary data from the study is expected in second-half 2022.
Mosunetuzumab is Roche’s investigational CD20xCD3 T-cell engaging bispecific antibody. The company has entered into a clinical supply agreement with Roche for the bispecific cohort of the EZH-1501 study.
A potential label expansion in any of the above indications should boost Tazverik’s sales in the days ahead. It also remains to be seen whether it can drive the stock in 2022 or not.
Apart from tazemetostat, Epizyme is evaluating its novel first-in-class oral SETD2 inhibitor, EZM0414, across relapsed/refractory multiple myeloma and diffuse large B-cell lymphoma in a phase I/Ib study (SET-101). Preliminary data from the study is expected in second-half 2022.
While Tazverik shows promise, Epizyme remains primarily reliant on a single drug for growth. An unfavorable outcome from any of the aforementioned development programs could adversely impact the company’s prospects and the stock’s performance. Lack of a strong pipeline beyond Tazverik also remains an overhang.
Epizyme, Inc. Price
Epizyme, Inc. price | Epizyme, Inc. Quote
Zacks Rank & Stocks to Consider
Epizyme currently carries a Zacks Rank #3 (Hold). Better-ranked stocks in the biotech sector are Athersys, Inc. ATHX and Kaleido Biosciences, Inc. KLDO, both carrying a Zacks Rank #2 (Buy) at present. You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Athersys’ earnings estimates have been revised 21.7% upward for 2022 over the past 60 days.
Earnings of Athersys have surpassed estimates in one of the trailing four quarters and missed the same on the other three occasions.
Kaleido Biosciences’ loss per share estimates have narrowed 23% for 2022 over the past 60 days.
Earnings of Kaleido Biosciences have surpassed estimates in three of the trailing four quarters and missed the same on the other occasion.
5 Stocks Set to Double
Each was handpicked by a Zacks expert as the #1 favorite stock to gain +100% or more in 2021. Previous recommendations have soared +143.0%, +175.9%, +498.3% and +673.0%.
Most of the stocks in this report are flying under Wall Street radar, which provides a great opportunity to get in on the ground floor.
Today, See These 5 Potential Home Runs >>Want the latest recommendations from Zacks Investment Research? Today, you can download 7 Best Stocks for the Next 30 Days. Click to get this free report
Roche Holding AG (RHHBY): Free Stock Analysis Report
Athersys, Inc. (ATHX): Free Stock Analysis Report
Epizyme, Inc. (EPZM): Free Stock Analysis Report
Kaleido Biosciences, Inc. (KLDO): Free Stock Analysis Report
To read this article on Zacks.com click here.
Zacks Investment Research
The views and opinions expressed herein are the views and opinions of the author and do not necessarily reflect those of Nasdaq, Inc.



